Optimization and validation of PCR for three hypervariable regions (HVI, HVII and HVIII) in human mitochondrial DNA
Shakeela Daud1*, Saqib Shahzad2, Muhammad Shafique1, Munir Ahmad Bhinder3, Muhammad Niaz1, Asif Naeem1, Azam Ali2, Zia-ur-Rehman1, Tayyab Husnain1
Adv. life sci., vol. 1, no. 3, pp.165-170, May 2014
*- Corresponding Author: Shakeela Daud (Email: imshakeela@yahoo.com)
Author Affiliations[Date Received: 14/05/2014; Date Revised: 23/05/2014; Date Published Online: 25/05/2014]
Abstract![]()
Introduction
Methods
Results
Discussion
References
Abstract
Background: mtDNA testing is often successful in cases where nuclear DNA is highly degraded or the sample source is too limited. Sequence polymorphism of D-loop region of mtDNA has been used for identification of forensic remains, analysis of mother–child relationships and comparisons between ethnic groups through maternal lineages. PCR conditions were optimized and validated for three hypervariable regions (HVR I- 480 bp, HVR II- 420 bp and HVIII- 255 bp) of mitochondria conducted at National Centre of Excellence in Molecular Biology, University of the Punjab – Lahore, Pakistan.
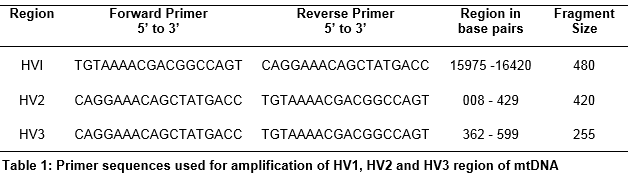
Methods: Blood samples of 86 individuals were drawn from 25 Pakistani families. DNA was extracted and purified by Sambrook method. DNA was quantified at agarose gel electrophoresis and N-D 1000 Nanodrop spectrophotometer. Three hypervariable regions (HVR I, HVR II and HVIII) of mitochondrial DNA were optimized with different PCR components and PCR conditions using three pairs of oligonucleotides along with reagent blanks, positive and negative controls.
Results: The best results for amplification were shown at 1x PCR buffer, 2.5mM Mgcl2, 0.3µl of Taq DNA polymerase (5u/µl), 0.2mM dNTPs, 0.8µM forward-reverse primers for HVR I, 0.7µM forward-reverse primers for HVR II and 0.4µM forward-reverse primers for HVR III at 52◦C annealing temperature.
Conclusion: Optimized PCR protocol for three hypervariable mtDNA regions has provided a way out to lead mtDNA analysis which is very necessary tool in those forensic biological samples, where nuclear DNA is highly degraded, to identify missing persons and determine maternal lineages.
Keywords: Mitochondrial DNA, Forensic analysis, D-loop region, Hypervariable regions
Introduction
Human mitochondrial DNA (mtDNA) is double stranded, circular molecule containing 16,569 base pair [1]. mtDNA is less susceptible to nucleases that have a tendency to degrade the DNA [2]. The circular mitochondrial DNA consists of highly polymorphic control region also known as D- loop region, is highly polymorphic having three hypervariable regions HVI, HVII and HVIII [3]. It encodes 2 rRNAs, 22 tRNAs and 13 polypeptides of which seven are components of complex I (NADH dehydrogenase), three are components of complex IV (cytochrome c oxidase), two are subunits of complex V (ATP synthase) and cytochrome b (a subunit of complex III). Most of this information is encoded in heavy (purine-rich) strand (02 rRNAs, 14 tRNAs and 12 polypeptides). The light (pyrimidine-rich) strand contains genetic information. Mitochondrial genes have no introns. Intergenic sequences are absent or limited to a few bases. Mitochondrial DNA has found a vital niche in forensic DNA testing, and its use is expanding very rapidly both in terms of casework and the number of laboratories performing mtDNA analysis [4]. Mitochondrial DNA sequence analysis gives the possibility of identifying victims of crime cases or missing persons where bodies remain undiscovered for many years [5]. In such cases, polymorphism of mtDNA can be obtained by sequencing which can be used for individual identification purposes like in missing persons, mother–child relationships and comparisons between ethnic groups by determining their maternal lineages [6].
Analysis of mtDNA is based on detection by Sanger’s sequencing of SNP variation in two most variable regions in the genome, HVI and HVII. Mitochondrial genetics is also different from Mendelian genetics on happening of its uniparental inheritance [7]. At present, two most important mtDNA analysis guidelines are being used in forensic science are (SWGDAM), the Scientific Working Group of DNA Analysis Methods of the FBI and the (EMPOP) mitochondrial DNA control region database [8].
Forensic DNA analysis is powerful tool in crime investigation however in some cases the analysis of genomic DNA fails due to limited or degraded template. In these cases sequence polymorphism of mitochondrial genome can serve as useful target [9]. The present work will also help to establish a PCR protocol leading to the identification of missing persons and also can make a relationship of those individuals with their families on the basis of mitochondrial DNA analysis.
Methods
Blood samples of 86 individuals from 25 Pakistani families were collected in 0.5M EDTA containing tubes with volunteer’s consent, the approval was given by the ethical committee of National Centre of Excellence in Molecular Biology, University of the Punjab, Lahore Pakistan. Mitochondrial DNA was extracted and purified by Sambrook method [10]. The quantitation of Mitochondrial DNA was performed by gel electrophoresis and N-D 1000 Nanodrop spectrophotometer. D-loop region, comprising three hypervariable regions HVI, HVII and HVIII of mitochondrial DNA, was optimized by using three pairs of primers shown in table 1. The sequence of primers was synthesized from DNA sequencing lab of National Centre of Excellence in Molecular Biology, University of the Punjab, Lahore Pakistan. The primers were optimized at variable concentrations of 10x PCR buffer (fermentas), 25mM MgCl2 (fermentas), 0.5U/µl Taq DNA polymerase (fermentas), 1mM dNTPs (fermentas), primers and mtDNA template at different annealing temperatures on ABI9700 PCR (Applied Biosystems Foster City, CA).
Results
PCR amplification conditions were optimized for each HV regions (I, II and III) of mitochondrial DNA) separately. The PCR reactions and PCR conditions were varied in MgCl2 ions from 1.5mM to 3mM working concentration, 0.5x to 2x PCR buffer (pH 8.0), 0.1mM to 0.2mM dNTPs concentrations, 0.5ng to 5ng mtDNA template and annealing temperatures ranging from 50°C to 57°C for these HV regions in 25µl reaction volume. Forward and reverse primer of 10pmol was used in concentration of 0.1µM to 1µM.
Amplification of PCR products were observed on agarose gel electrophoresis and amplification results of approximately 480 bp band for HVI region of mtDNA which were visualized under UV light as shown in figure 2 along with ladder (100bp-1000bp). Reagent blank and negative controls were used to detect the contamination in extraction and PCR solutions respectively which showed no amplification. Using the variable concentrations of PCR components HVII region of mitochondrial DNA was amplified and the best results shown in figure 3 compared with ladder represents on agarose gel image of the amplification product of 420 bp of mitochondrial DNA fragment. HVIII region of Mitochondrial DNA was also amplified with same PCR conditions and fragment of 255 bp long was obtained according to the ladder as shown in figure 4.
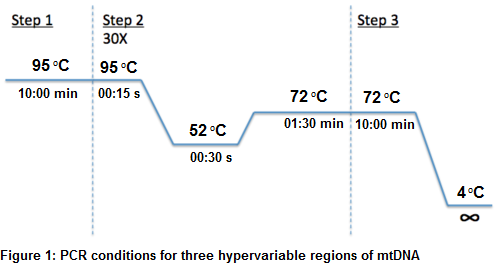
The best conditions for amplification for all three hypervariable regions were Obtained with 1x PCR buffer, 2.5mM Mgcl2, 0.3µl of Taq DNA polymerase (5U/µl), 0.2mM dNTPs, 0.4µM forward-reverse primers for HVR I, 0.7µM forward-reverse primers for HVR II and 0.8µM forward-reverse primers for HVR III at 52◦C annealing temperature with 30 PCR run cycles as shown in figure 1 using 2ng of mtDNA template in 25µl PCR reaction volume.
Figures
Discussion
Human mtDNA, a small (16,569bp) circular genome containing 37 genes is found in cellular cytoplasm containing a noncoding region of 1100bp known as D-loop or control region [11]. During the process of optimization, conditions for PCR reaction, optimal quantity of template DNA, Taq DNA polymerase, annealing temperature, pH and Mgcl2 concentration were specified.
Optimal quantity of required mtDNA was also obtained by using different concentration from 0.5ng to 5ng but amplification of the regions HVI, HVII and HVIII was obtained with 2ng of DNA according to optimal concentrations of the other PCR components. PCR reaction without proper quantification of template DNA may not produce sufficient amplicon quantity to be analyzed properly. Optimization of PCR amplification plays an important role in mtDNA sequencing. 10X PCR buffer was used in PCR optimization. Different concentrations of MgCl2 ranging from 1.5mM, 2mM, 2.5mM, 3.0mM and 3.5 mM. Best results were observed on 2.5mM Mgcl2 level. Range of Taq DNA polymerase (5 units) quantity used from 0.3µl to 0.5µl for PCR amplification reaction. Best results were observed on 0.3µl from 5 unit of Taq DNA polymerase using 1X PCR buffer in the final reaction volume. Different number of amplification cycles were applied, it was observed that 30 cycles produced good results. The PCR conditions applied by Hoong and Lek were initial denaturation at 94ºC for 5min, followed by 94ºC for 45 seconds, 66ºC for 1 min, 72ºC for 1 min, 72ºC for 5min with 35 cycles but amplification results also obtained with PCR conditions as initial denaturation at 95ºC for 10 min, followed by 95ºC for 15 seconds, 52ºC for 30 seconds, 72ºC for 1:30 min, 72ºC for 10 min in 30 PCR cycles [12].
The conditions optimized for three hypervariable regions of mitochondria may also be used in the future for sequence polymorphism of three hypervariable regions of mtDNA to analyze highly degraded forensic samples, to identify missing persons and to determine maternal lineages.
References
-
Anderson S, Bankier AT, Barrell BG, De Bruijn M, Coulson AR, et al. Sequence and organization of the human mitochondrial genome. (1981).
-
Ilyas M, Israr M, ur Rahman Z. Copy Number Variation in Forensic Science. Advancements in Life Sciences, (2014); 1(2): 1-2.
-
Date Chong M, Calloway C, Klein S, Orrego C, Buoncristiani M. Optimization of a duplex amplification and sequencing strategy for the HVI/HVII regions of human mitochondrial DNA for forensic casework. Forensic science international, (2005); 154(2-3): 137-148.
-
Schneider PM, Bender K, Mayr WR, Parson W, Hoste B, et al. STR analysis of artificially degraded DNA—results of a collaborative European exercise. Forensic science international, (2004); 139(2): 123-134.
-
Bender K, Schneider PM, Rittner C. Application of mtDNA sequence analysis in forensic casework for the identification of human remains. Forensic science international, (2000); 113(1): 103-107.
-
Lima G, Pontes M, Abrantes D, Cainé L, Pereira M, et al. HVI and HVII sequence polymorphisms of the human mtDNA in the North of Portugal: Population data and maternal lineages; 2006. Elsevier. pp. 133-135.
-
Taylor RW, Turnbull DM. Mitochondrial DNA mutations in human disease. Nature Reviews Genetics, (2005); 6(5): 389-402.
-
Parsons TJ, Coble MD. Increasing the forensic discrimination of mitochondrial DNA testing through analysis of the entire mitochondrial DNA genome. Croatian medical journal, (2001); 42(3): 304-309.
-
Salas A, Lareu V, Calafell F, Bertranpetit J, Carracedo A. mtDNA hypervariable region II (HVII) sequences in human evolution studies. European Journal of Human Genetics, (2000); 8(12).
-
Sambrook J, Fritsch EF, Maniatis T Molecular cloning. Chapter: Book Name. 1989 of publication; 2; Cold spring harbor laboratory press New York.
-
Chen M-H, Lee H-M, Tzen C-Y. Polymorphism and heteroplasmy of mitochondrial DNA in the D-loop region in Taiwanese. JOURNAL-FORMOSAN MEDICAL ASSOCIATION, (2002); 101(4): 268-276.
-
Hoong LL, Lek KC. Genetic polymorphisms in mitochondrial DNA hypervariable regions I, II and III of the Malaysian population. Asia-Pacific Journal of Molecular Biology and Biotechnology, (2005); 13(2): 79-85.


![[]Figure 2](http://www.als-journal.com/wp-content/uploads/2014/05/Figure-2.png)
![[]Figure 3](http://www.als-journal.com/wp-content/uploads/2014/05/Figure-31.png)
![[]Figure 4](http://www.als-journal.com/wp-content/uploads/2014/05/Figure-41.png)