Short Communication
Protocol optimization for efficient in vitro micro-propagation of Stevia rebaudiana Bertoni
Muhammad Aslam Javed, Imran Habib*, Muhammad Waqas Jamil, Muhammad Jamshaid Anwar, Shahid Nazir, Sajid-ur-Rahman, Muhammad Zaffar Iqbal
Adv. life sci., vol. 6, no. 3, pp. 100-105, May 2019
*– Corresponding Author: Imran Habib (Email: imranuaf@gmail.com)
Authors' Affiliations
Abstract![]()
Introduction
Methods
Results
Discussion
References
Abstract
Background: Stevia rebaudiana Bertoni is one of the most amazing herb that is an abundant source of diterpene glycoside a non-caloric sweetener. In recent years alongside its utilization in herbal and folk medicine, commercial exploitation of this herb for weight control and anti-diabetic treatment has started.
Methods: In present study healthy nodal segments of Stevia rebaudiana Bertoni were used as explants for optimization of in vitro micro-propagation and regeneration protocol for rapid, fast and efficient multiplication under in-vitro conditions. For shoot proliferation and rooting various concentrations cytokinins NAA (Naphthalene acetic acid), BAP (Benzyl amino purine), IAA (Indole acetic acid) and kinetin (Kn) were employed.
Results: Shoots without roots were regenerated from nodal segments via auxiliary shoot induction. The production of multiple shoots from nodal portion was maximum on MS medium complemented with 0.5 mg/L of BAP and 1.5 mg/L of Kn. Maximum rooting was recorded on MS medium supplemented with 0.25 mg/L NAA and 1.0 mg/L of IAA. Successful hardening was achieved under controlled growth room conditions followed by transfer to glass house and open field.
Conclusion: Standardization of explant source and culturing media for multiplication of shoot resulted in mass proliferation of homogenous and healthy plantlets of Stevia clones under local conditions.
Keywords: Stevia rebaudiana Bertoni; BAP; Kinetin, NAA; IAA; Micro-propagation; Regeneration
Stevia rebaudiana is by nature a perennial plant that belong to family Asteraceae. The leaves of this shrub are naturally sweet and according to some studies contain up to 300 times more sweeteners than sugarcane [1]. The source of natural sugar is a glycoside known as stevioside. Due to absence of calories in this stevia-based sweetener, it is medically prescribed for patients with diabetes and excessive weight problem [2]. Stevioside also hold antibacterial properties and known to inhibit the growth of Staphylococcus, Bacillus subtilis and E. coli bacteria [3]. Owing to these antibacterial abilities the leaf extract of stevia is used in the treatment of open wounds and sore gum disease [4].
Stevia plant can be propagated by seed and shoot cuttings, however, seed grown plants show poor germination and undesirable changes in stevioside levels and composition [5]. Under such circumstances in vitro micropropagation of S. rebaudiana seems to be only viable method to multiply and to overcome the stevioside variability and seed production problems.
Although several tissue culturing protocols of various varieties and clones of S. rebaudiana has been reported in literature. It is of utmost importance to adopt the micropropagation procedure that is specific to genetic material available for a particular region [6]. There are also diverse factors that significantly affect the regeneration of shoot that include source of explant, tissue sterilization practices, media preparation, culturing conditions and their optimizations, production of phenolic compounds and discoloration of media and even use of diverse genotypes of the same species of Stevia in culturing experiments [7].
Hence, the present study was undertaken to investigate and standardize an effective, economical and simple in vitro regeneration protocols for Stevia rebaudiana Bertoni under local environmental conditions.
Nodal segments of Stevia rebaudiana Bertoni were collected form Agricultural Biotechnology Research Institute (ABRI), Faisalabad and used as explants for in vitro micropropagation and regeneration. The present research was undertaken at the Center of Agricultural Biochemistry and Biotechnology (CABB), University of Agriculture (UAF), Faisalabad.
The explants were thoroughly rinsed under clean tap water to remove dust and soil particles. Supplementary washing was given by 5% (w/v) sodium hypochloride (NaOCl) solution for 2-3 minutes followed by surface sterilized using 70% ethanol for 1 minute and final rinsing was performed 5 times with autoclaved d3H2O. Additional sterilization was done by using exposing the tissues to UV light in air laminar flow for providing optimum sterilized conditions in working area.
MS basal medium [8] was used throughout the experiment as nutrient medium and supplemented with iron, organic supplements, carbon source and vitamins was used to culture the nodal explants. The pH of nutrient media was maintained at 5.75–5.80 prior to autoclaving by adding 1N HCL or 1N NaOH. Different concentrations (0.00-3.00 mg/L cytokinins and 0.00-2.5 mg/L auxins) of plant growth promoting hormones alongwith MS basal medium were also used. Cultured explants were incubated at 25 ± 2°C under growth-room conditions with a period of 16 h light and 8 h dark period with light intensity of 25 μ M m-2 s-1. Plant growth hormones employed in the tissue culture media were auxins and cytokinins. Auxins hormones were IAA and NAA and cytokinins were BAP and Kn, respectively. Being thermostable, these hormones were incorporated in the nutrient media prior to autoclaving. Various concentrations of sucrose or glucose (25-30 g/L) in the MS basal media were used as carbon source.
For shoot proliferation sterile nodal segments of Stevia were inoculated on MS basal media complemented with constant dose of 0.5 mg/L BAP in various combinations with Kn (i.e. 0.0, 0.5, 1.0, 1.5, 2.0 and 2.5 mg/L). Another set of nodal segments was checked for optimum multiple shoot regeneration by supplementing MS basal nutrient media with different levels of BAP (i.e. 0.0, 0.5, 1.0, 1.5, 2.0, 2.5, 3.0 mg/L) having constant concentration of Kn (i.e. 1.5 mg/L).
Regenerated rootless shoots were utilized as explants for in vitro root induction. Plantlets of approximately 4-5 cm length were excised and inoculated on MS basal media complemented with various concentrations of NAA (i.e. 0.00, 0.25, 0.50, 0.75, 1.00 and 1.50 mg/L) and IAA (0.0, 0.5, 1.0, 1.5, 2.0 and 2.5 mg/L), respectively.
Rooted plantlets were extracted from the test tubes and roots were gently washed with sterile distilled water to remove the nutrient media. The plantlets were then shifted to dark colored poly-planter bags filled with sterilized soil and sand mixture. To conserved moisture, plantlets were covered with clear polythene bags, placed in the incubation room at 26 ± 2°C, with 70-80 % relative humidity and under luminance of 0.089 μMm-2s-1. The plastic bags were gradually removed from the top of the plantlets for three weeks and after that placed at temperature of 25°C for another two weeks. Finally, for hardening the plantlets were transferred to the field and kept under partial shade for 4 weeks to avoid direct sunlight. Growth data of the plantlets were recorded regularly after 15 and 30 days of interval on the basis of number of multiple shoot formation, shoot length, number of roots and leaves, root length, numbers of nodes and survival percentage of plants during acclimatization and analyzed.
For statistical analysis, Complete Randomized Design was applied by using Minitab v17 statistical software.
Nodal sections having 2 to 3 nodes (Fig. 1A) were placed on shoot induction media. MS nutrient media complemented with different concentrations of BAP and Kn (0.0, 0.5, 1.0, 1.5, 2.0, 2.5 and 3.0 mg/L) was evaluated in combination with each other and also in individual form.
Shoot induction
All treatments induced different percentages of shoot proliferation which differed in length and number of shoots per explant. To measure the growth response of Stevia plantlets data regarding various parameters like %age of buds response, number of shoots initiated/explants and shoots length was recorded. Buds started responding by bud break and shoots appeared after 15 days of culture. Further, shoots were separated and transferred to different multiplication media. Number of experiments were carried out to optimize the swift multiplication of shoot. These included incorporation of high concentration of cytokinins, (i.e. 0.0, 0.5, 1.0, 1.5, 2.0, 2.5 and 3.0 mg/L of BAP and 0.0, 0.5, 1.0, 1.5 and 2.0 mg/L of Kinetin) in the media. A difference (about 1.5 cm) between shoots induction was observed on various concentrations of BAP and kinetin (Fig. 1B). In present study BAP and Kn were evaluated either in combined or in separate form.
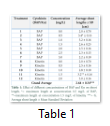
Table 1 shows an average length at each concentration of cytokinins to induce shoot proliferation. Average shoot lengths at all treatments was about 2.64 cm. Considering performance of individual cytokinin in the media, 0.5 mg/L BAP with 3.4 cm average shoot length was found to be the best dose for shoot elongation whereas the optimum dose of Kinetin was 1.5 mg/L at which average shoot length of 3.2 cm was observed (Table 1). Uniform plants in large number were generated and they were shifted to shoot multiplication medium to achieve the optimum shoot length and number of shoot/explants.
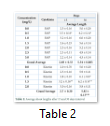
Average shoot length after 15 and 30 days intervals were recorded for two major cytokinins (BAP and Kn). Here 7 treatments of BAP (ranging from 0.0 to 3.0 mg/L) and 5 treatments of Kn (ranging from 0.0 to 2.0 mg/L) were evaluated for finding the best performing dose for multiple time duration. Keen observations showed that maximum average shoot elongation was achieved when 0.5 mg/L BAP was provided in the media for 30 days. However, in case of Kn almost same average shoot length was observed after the same time period of sub-culturing (Table 2).
Shoots multiplication
Successfully generated Stevia plantlets without root were exposed to different treatments of combined concentrations of cytokinins (i.e. BAP and Kn) supplemented with MS nutrient media to boost multiplication of regenerated stems. Concentrations of BAP and Kn were managed within the range of 0 to 3 mg/L (i.e. 0.0, 0.5, 1.0, 1.5, 2.0, 2.5, 3.0 mg/L).
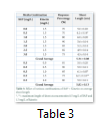
After 6-8 weeks of sub-culturing, length and number of shoots/explants were recorded at various combinations of BAP and Kn (Fig.1C). When Kn was kept constant with a dose of 1.5 mg/L, its combination with 0.5 mg/L BAP was found to be ideal for attaining maximum shoot length of 6.2 cm. Not surprisingly same results were observed when at a constant dose of 0.5 mg/L of BAP was provided in the media with Kn concentration of 1.5 mg/L thus proving the effectiveness of this Plant Growth Regulator (PGR) combination. At this level of PGRs healthy dark green colored leaves and stem were observed. Average shoot length was 5.34 and 5.82 cm and number of shoots per explant were 3.50, respectively (Table 3).
Root induction
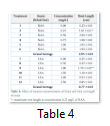
Rooting media was prepared under aseptic conditions and plantlets having no root were shifted on this media. Table 3 represents various rooting media composed of 4.33 g/L MS nutrient media supplemented with different concentrations of both NAA and IAA in solitary form ranging from 0.0 to 2.50 mg/L (i.e. 0.0, 0.25, 0.50, 0.75, 1.0 and 1.50 of NAA and 0.0, 0.50, 1.0, 1.5, 2.0 and 2.5 mg/L of IAA, respectively).
Length and number of roots differed due to varying concentrations of auxins (Fig. 2A, 2B). Maximum length of root was observed at the individual concentration of 0.25 mg/L of NAA (3.30 cm) and 1.0 mg/L of IAA (1.70 cm), respectively (Table 4). Root lengths and number gradually decreased with an increase in concentration of IAA and NAA. Considering the average root length for all NAA and IAA treatments, highest root length mean of 2.05 cm was recorded in case of NAA application whereas IAA was far less affective in this regard with 0.77 cm root length (Table 4).
Hardening
Completely established stevia plantlets having 90% survival rate were shifted to containers filled with sand, soil and peat in equal quantity (i.e. 1:1:1, respectively). Plantlets lacked frost and low temperature resistance in the open environment. Further, it was observed that lack of humidity also effected the survival rate of plantlets in the open environment. Therefore, the stevia plants were enveloped with transparent polythene sheet and kept at 23 to 25o C in incubation room for 2 to 3 weeks. Established and vigorous plants were shifted carefully into larger containers and were kept in fields under shade for 9 to 10 days. Finally, fully developed plants were transplanted in the field as shown in Figure 2C.
Figures & Tables
BAP and Kn although are two separate cytokinins used for shoot initiation purpose. However, both these PGRs individually as well as in combination showed efficient plant growth and development properties when nodal sections of Stevia were used as explants.
It was observed that BAP seemed to promote improved proliferation than that of Kn because it could induce shoot proliferation in all three explants while Kn was useful to encourage proliferation only for shoot apex and nodal stem sections [10]. However when explants of cucumber were cultured on various cytokinins, MS medium containing Kn was found to the most effective for encouraging shoots from nodal parts. The highest regeneration of 83% and maximum shoot length (3.61 cm) was observed on MS media combined with 1.0 mg/L Kn. Interestingly least culture response was documented in case of media fortified with either BAP or Zeatin [11]. Abdul-Razak et al. reported similar findings while investigating the optimum media for micropropagation of Stevia in Malaysia [12]. Highest shoot formation of 7.82 shoots per explant was obtained on a MS media supplemented with 0.5 mg/L BAP and 0.25 mg/L Kn. In contrary to this study our results are in complete agreement with the findings of [13] who reported prolific shoot elongation from nodal shoot meristems of Passiflora foetida L. on semi solid MS media having 2.0 mg/L BAP. They reported 16.45 ± 0.44 shoots per explant when further culturing was done on MS media supplemented with 0.5 mg/L each of BAP and Kn.
Forcing the root induction is the most critical and difficult part of any micropropagation experiment and there is no exception in case of Stevia plant. For this purpose various auxins like IAA and NAA have been utilized in various studies [14-17]. Maximum average length of root (3.30 cm) was observed when an individual dose of 0.25 mg/L of NAA was supplemented in MS media having full strength. These results are in complete agreement with the observation of [15] where he reported a higher concentration of 2.0 mg/L NAA to the best auxin dose that induced 90% root induction in a Medicinal shrub, Holarrhena antidysenterica. However, in a contradictory study when segments of C. officinalis seedlings were cultured on Murashige and Skoog (MS ½ strength) medium, about 80% higher rate of rooting from the apical explants was obtained at low level of IAA i.e. 0.1mg/L [16]. The effectiveness of NAA as a rooting hormone was also proved by [17] who micro-propagated two commercial varieties of sugarcane on media containing NAA and IBA. The results showed that 1.0 mg/L NAA was the optimum concentration at which maximum roots were observed.
Standardization of explant source and culturing media for multiplication of shoot resulted in mass proliferation of homogenous and healthy plantlets of Stevia clones under local conditions. This experiment demonstrated an alternative propagation method to enhance the production of Stevia at commercial scale in comparison to conventional multiplication techniques.
The authors declare that there is no conflict of interest regarding the publication of this paper.
- Abdul-Razak UN, Ong CB, Yu TS and Lau LK. In vitro Micropropagation of Stevia rebaudiana Bertoni in Malaysia. Braz. Arch. Biol. Technol., (2014); 57 (1): 23-28.
- Shekhawat MS, Kannan N, Manokari M, Ravindran CP. In vitro regeneration of shoots and ex vitro rooting of an important medicinal plant Passiflora foetida L. through nodal segment cultures. Journal of Genetic Engineering and Biotechnology, (2015); 13: 209-214.
- Sevik H. and Kerim G. Effects of IAA, IBA, NAA, and GA3 on Rooting and Morphological Features of Melissa officinalis L. Stem Cuttings. The Scientific World Journal, (2013), Article ID 909507, 5 pages. 10.1155/2013/909507
- Kanungo S, Pradhan C, Sahoo SL, and Sahu RK. Role of auxins in the in vitro rooting and micropropagation of Holarrhena antidysenterica Wall., a woody aromatic medicinal plant, through nodal explants from mature trees. Journal of Medicinal Plants Research, (2012); 6(31): 4660-4666.
- Victorio CP, Lage CLS, and Sato A. Tissue culture techniques in the proliferation of shoots and roots of Calendula officinalis. Revista Ciência Agronômica, (2012); 43 (3): 539-545
- Tolera B. Effects of Naphthalene Acetic Acid (NAA) and Indole -3- Butyric Acid (IBA) on In Vitro Rooting of Sugarcane (Saccharum officinarum L.) Micro-Shoots. Journal of Biotechnology and Biomaterials, (2016); 6: 215. doi:10.4172/2155-952X.1000215.
This work is licensed under a Creative Commons Attribution-Non Commercial 4.0 International License. To read the copy of this license please visit: https://creativecommons.org/licenses/by-nc/4.0